 Hydrogen bonds are usually showed as dotted lines between two atoms. This is a very weak bond and strength of hydrogen bond (5-10 Kcal per bond) is much less than the strength of covalent bond. Ionic bonds are not as strong as covalent, which determines their. (The statement is a fairly complete definition of the hydrogen bond Hydration shell refers to organization of water molecules around a non-polar group "Hydrophobic Bond" is not a real term, don't confuse it with a "hydrogen bond" Partial charges cannot be involved in ionic bonds, only full charges can form ionic bonds.)Ģ. Hydrogen bond is an attractive force between a partially positive charged hydrogen and a partially negative charged atom (oxygen and nitrogen). Ionic and covalent bonds between elements require energy to break. Micelles will form spontaneous driven by the hydrophobic effect.ġ. The partial negative charge at one end of a water molecule is attracted to the partial positive charge of another water molecule. Micelles are aggregates of amphipathic molecules that sequester the nonpolar part on the inside, much like the inside of an orange. Hydrogen bonds have considerable importance in biochemistry. These can form micelles if the nonpolar part is sufficiently large. An example is a fatty acid, which has a charged carboxylate (red) and a non-polar hydrocarbon chain (yellow). CompoundĪmphipathic (or amphiphilic) compounds are both polar (or charged) and nonpolar. 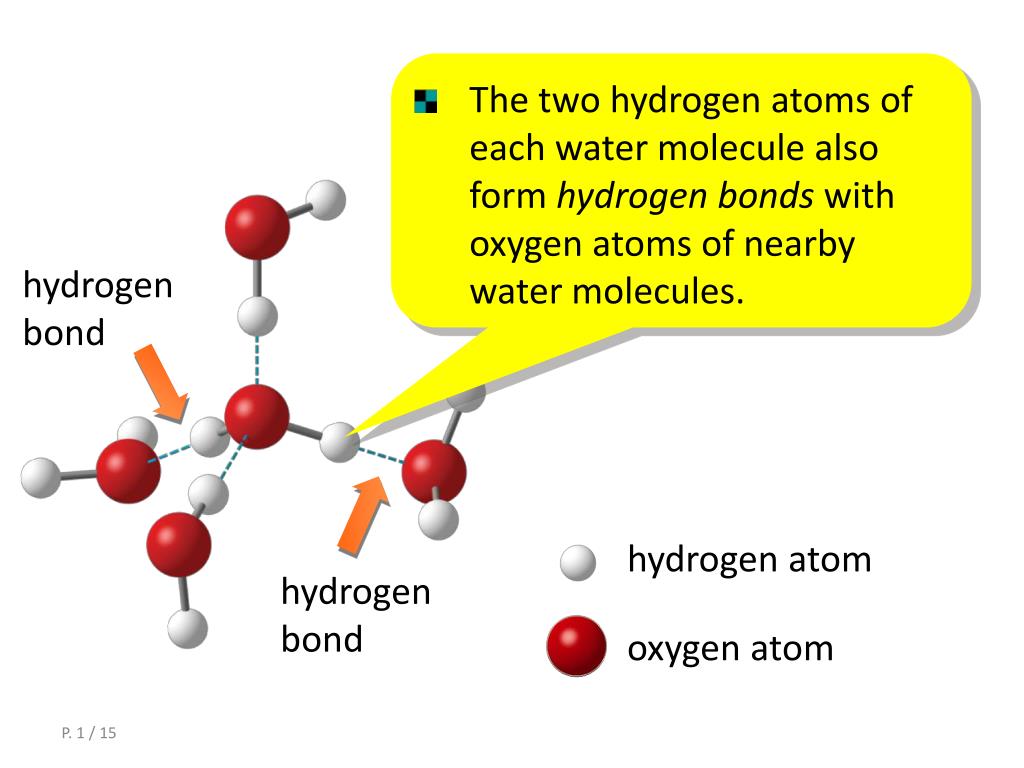
A large dipole moment means that the solvent molecules can interact favorably with charged solute molecules. The dielectric constant is proportional to the dipole moment of the solvent, as the dipole moment increases, D, increases, as shown in the following table. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
